About the Author

Marjolein Van Egmond
Professor at Amsterdam UMC
Marjolein van Egmond's laboratory studies antibody activation of innate myeloid immune cells, macrophages and neutrophils, with emphasis on understanding the role of immunoglobulin A (IgA) in physiology and pathology. Her groups research focuses specifically on (1) the contribution of abnormal antibody responses to chronic inflammation and autoimmunity - (2) monoclonal antibody therapy of cancer. Dr. van Egmond’s research is highly translational and she is cross appointment with the Department of Surgery and the Department of Molecular Cell Biology and Immunology, to facilitate the rapid progression of pre-clinical findings into clinical applications.
Her research is funded by The Dutch Cancer Society, Worldwide Cancer Research, MaagLeverDarm Foundation, Cancer Center Amsterdam Foundation and the Netherlands Organisation for Scientific Research (VENI; 2003, VIDI; 2007, ASPASIA; 2008, VICI; 2014, ASPASIA 2015).
Contents
This article was originally published on April 9 as a flipbook PDF, illustrated with playful illustrations. For those who find swiping and scrolling annoying, I quickly made a text-only version. The typography is good enough to take in the content. There are still illustration captions here and there, and there are also captions in the table of contents. I left it, with some imagination you might be able to think of something yourself and otherwise you will still look in the original article.
SARS-CoV-2 is a new virus for humans, and COVID-19, the disease it causes, is a new disease. Much is still unknown. How contagious is the virus in people of different ages and with different stages of the disease? What is the range of symptoms? Are there many people who hardly have any symptoms? How can the disease be treated? Scientific discoveries are moving fast and learning is happening every day. However, not everything is clear yet. What is told today may turn out to be slightly different tomorrow when new data have become known.
The situation is closely monitored on a daily basis and models are made as well as possible to predict the progress of the pandemic based on the most up-to-date scientific knowledge. These predictions will not always be completely correct. This makes sense. For these models, various assumptions must be made, such as the infectivity and lethality of the disease. The models also predict the effects of measures (such as staying at home and keeping a distance from each other) on the spread of the disease and the number of people who need to be admitted to hospital. When new knowledge is gained, it may turn out afterwards that some of these assumptions were not exactly correct.
The same applies to the measures taken by the government on the basis of these models. As Prime Minister Rutte has previously said, the government must 'make 100% percent decisions with only 50% information'.
The models help enormously in decision-making. Each new phase of the disease and the number of infections per country requires different measures from the population to keep the disease under control and not to overwhelm hospitals. That is why the policy is always adjusted when new information is available about the number of infections in the Netherlands.
This 'Corona issue' from the Dutch Society of Immunology describes the information about the SARS-CoV-2 virus and the immune system as it was known in early April 2020.
For the latest information on the SARS-CoV-2 virus and COVID-19:
For more information about vaccination:See https://rijksvaccinatieprogramma.nl/
For more information about the immune system: See https://mijnafweer.nl
The Coronavirus
A virus is not a cell, but consists of a bit of hereditary (or genetic) material surrounded by a shell of fats and proteins. They are microscopic particles, which are so small that they cannot be seen with an ordinary microscope, but only with a so-called electron microscope. On average, viruses are 100 times smaller than a bacterium. Coronaviruses owe their name to their appearance. They have regularly arranged protrusions, which make it look like they are wearing a crown (crown = corona in Latin). Viruses need a host to survive.
There are many different coronaviruses, which usually infect birds and mammals. Bats are remarkably often infected and a host for coronaviruses. The coronavirus that is causing the current pandemic is also thought to be a natural host of a bat, namely the horseshoe-nosed bat. It is possible that the virus jumped via the Javan pangolin, which then brought it to humans at the Huanan Seafood market in Wuhan (China), where the pangolin is traded.
The new virus is called SARS-CoV-2. The disease that causes this is called COVID-19
So far, six coronaviruses have been found that can infect humans. These include SARS and MERS, which were the cause of epidemics in 2003 and 2012. Because this new coronavirus is very similar to SARS (which is officially referred to as SARS-CoV), the official designation of the new virus is SARS-CoV-2. Coronaviruses generally infect the upper respiratory tract and the gastrointestinal tract. This is usually accompanied by mild symptoms, such as a cold, sore throat, cough, mild fever or diarrhea. Some variants, such as MERS, SARS-CoV, and SARS-CoV-2 also infect the lower respiratory tract (the lungs) and can cause life-threatening respiratory infections and pneumonia. The lung disease that causes SARS-CoV-2 is called COVID-19.
A virus depends on a host cell for multiplication
A virus can have DNA or RNA as genetic material, but is completely dependent on a host cell to replicate itself. Human cells contain DNA, the genetic code of life. The sequence of DNA determines which proteins are
produced. However, this does not happen directly, but through an intermediate step, the RNA. A virus can integrate its genetic material into human genetic material, regardless of whether it contains DNA or RNA.
Step 1 is the binding of a virus particle to a cell. To do this, the virus abuses a recognition molecule that is on the host cell. This is also called a receptor and has a completely different function in our cells. However, the virus has adapted (=mutated) in such a way that it can bind very specifically to the recognition molecule on the host cell. The protrusions of the SARS-CoV-2 virus bind to a recognition molecule (= ACE-2 receptor) that is found on cells in the lung and intestines, among other things. In this way, the virus can enter and hijack the cell.
Step 2 is the injection of the genetic material into the host cell. In the case of the SARS-CoV-2 virus, this is a single strand of RNA. The host cell mistakes this for its own RNA and starts multiplying it. Virus proteins are now also being made (step 3). From these new copies of the RNA and virus proteins, new virus particles are assembled. This usually leads to the death of the host cell. This releases the new viruses and infects nearby host cells, allowing the disease to develop exponentially.
Symptoms of infection with SARS-CoV-2 are highly variable
Symptoms occur on average between five days and two weeks after infection. The time that someone is already infected, but not yet sick, is called the incubation period. The symptoms of infection with the SARS-CoV-2 virus are very variable. So far, it seems that at least eight out of ten people have mild symptoms, such as dry cough, sore throat and/or fever, fatigue and muscle pain. Intestinal complaints such as nausea and diarrhea can occur and some sick people have a lot of headaches. Usually the symptoms are over within five days and people get better on their own. There are also people who have no or hardly any complaints. There are indications that this group is larger than thought, but that is difficult to prove at the moment due to a lack of testing.
So it may be that people with very mild symptoms do not know that they are infected. As a result, they are not tested and their number is not included in the RIVM counts. About two in ten patients develop serious complaints, such as shortness of breath and pneumonia. This usually only happens after about six days of the disease. It is therefore important to pay close attention to whether a sick person becomes more or less ill during this period. The condition of about a quarter of these patients can deteriorate so much that they have to go to hospital. This happens, among other things, when patients above 40 degrees get a fever or become very short of breath. The situation can even become critical for some patients, because they develop serious breathing problems. This is called acute respiratory distress syndrome (ARDS). These patients must be admitted to an intensive care unit (ICU), where they must be ventilated. Ventilation can take up to three weeks for patients to slowly get better. Unfortunately, a number of people also die from the disease.
The mortality rate depends on characteristics of the virus and other factors
It is difficult to determine how deadly the virus is while the pandemic is still going on. The mortality rate (in official terms case-fatality ratio (CFR)) is also not the same in all countries. In part, this is because it is not known exactly how many people really have the infection. Some countries test a lot, while other countries, such as the Netherlands, mainly test patients who are so sick that they come to the hospital. If it is not known how many people have become infected in total, the mortality rate cannot be calculated. According to the latest estimates, the mortality rate (or CFR) is about 1.2%. This would mean that about one in a hundred infected people dies from the infection. This is probably an overestimate, because the people who have no or mild symptoms and do not know that they are infected with the SARS-CoV-2 virus are not counted. At the moment it is unclear how many people this concerns, which makes all calculations and estimates difficult.
In addition to the properties of the virus, its lethality also depends on other factors. The composition and health of a country's population before the outbreak have a major impact. Older people and people who have other chronic health problems, such as cardiovascular disease, diabetes or respiratory problems, are particularly likely to die from COVID-19. People with obesity are likely to have a greater risk of ICU admission, while children and young people have a lower risk of death, although they can become (seriously) ill. Unfortunately, we see many people in the age of 30-50 years in hospital admissions and ICU admissions.
In addition, the quality and capacity of healthcare in a country determines the outcome. The mortality rate will be lower in a country with good health care and relatively young healthy people. It is also very important that there is no situation in which hospitals are overwhelmed with patients, so that care is no longer optimal. The latter is already a problem in several countries and is a major threat to all countries.
The SARS-CoV-2 virus is mainly transmitted from one person to another. The virus is released into the air through small droplets, by coughing and sneezing from an infected person, and when other people inhale those droplets, they can become infected with the virus. In addition, the virus can end up in the mouth, nose or eyes through the hands. The sicker someone is, the more virus someone can spread by coughing and sneezing a lot. However, this new virus also seems to be able to spread when people do not yet have so many symptoms. Now that it is becoming clearer that there is a (large) group of people who have no symptoms, but unknowingly spread the disease, this seems to be an additional explanation for why the pandemic is spreading so quickly. But how large this group is (the latest estimates suggest 25%) and what its exact contribution is to the spread still needs to be investigated. It is also unclear what the contribution is of the virus that is on objects. It is clear that depending on the material (paper, plastic, metal) the virus can be present for hours to days.
The multiplication number R indicates the infectivity of a virus
Virologists indicate the infectiousness of a virus with a multiplication number. At the beginning of the outbreak, this is called R0 and in SARS-CoV-2 it is estimated at 2.2. This means that each infected person infects an average of 2.2 other people. The R0 value is based on the initial situation when no measures have yet been taken and everyone is susceptible. During the outbreak, this number can change and is no longer called R0, but Rt, which stands for the effective infection number. When the R0 or Rt is greater than 1, the number of
Cases of infection are increasing exponentially. The higher the R, the faster the growth. For example, if R is 2, each person infects 2 others, who again infect 2 people (from 2 to 4
to 8 to 16 and so on). At 3 it goes much faster (3, 9, 27, 81....). With an R lower than 1, the infection will slowly fade out. It is therefore important to take measures, such as washing hands, keeping your distance and staying at home so that the increase number goes down.
Due to the measures taken, R goes down
In the Netherlands, these measures seem to be having an effect and the Rt is falling. The RIVM published on 1 April 2020 that the Rt has dropped below 1 in a few weeks. That is hopeful. Because people may have been infected earlier, it is possible that many people will still have to be admitted to hospital in the short term, or that many people will die. But hopefully those numbers will also go down in the short term. In any case, it is already the case that the number of hospital admissions is no longer increasing as quickly as at the beginning of the pandemic. It is important to keep up the measures for the time being, because otherwise the number of infections could increase quickly.
Keep your distance and stay at home as much as possible
The main route of infection between people is via droplets containing virus particles that have been coughed out or sneezed out. The best way to reduce this risk is to keep a distance from each other (at least 1.5 meters) and avoid contact. That is why people should stay at home when they have cold symptoms such as sneezing, sore throat, coughing or fever. But healthy people should also work from home as much as possible and avoid social contact with others, especially when someone falls into a risk group (the elderly and/or people with a chronic illness).
There is a lot of discussion about the usefulness and necessity of face masks. When someone is infected, face masks could prevent the spread of droplets. Wearing a face mask yourself to avoid becoming infected has less effect - depending on the type of face mask. Paper face masks in particular do not close properly and can let virus particles through. In addition, contamination can also occur through the eyes. Even professional face masks can actually increase the risk of contamination if they are not used properly. That is why in the Netherlands, only medical staff are currently advised to wear face masks.
Washing hands with soap lowers the risk of contamination
When an infected person sneezes or coughs, virus particles can get on their own hands as well as on door handles, faucets, shopping carts, and other objects. That is why the advice is to cough or sneeze into the elbow, not to shake hands and to wash hands often with soap. Touching the face with hands with virus particles can also cause infection. It is best to wash hands with soap, as this dissolves the fats of the virus and the virus falls apart. It is important to wash your hands long enough (at least 20 seconds) so that the soap can work properly. All parts of the hands (including nails) and wrists must also be washed thoroughly.
The immune system is like an army
The lymph nodes are the barracks, the blood vessels and the lymphatic vessels are the pathways that connect them to each other.
The immune system is a collective term for an extensive defense system that consists of many different molecules, immune cells and tissues. It can be compared to an army. An army consists of different parts, in which everyone has a specific function and task. Many immune cells are collected in special places in the body, such as in the lymph nodes, spleen and bone marrow. This can be compared to barracks. Just as barracks are connected by a road network, over which the troops can move, the lymph nodes are in contact with each other through the lymphatic vessels. The blood vessels are a second pathway network, which is also in contact with the lymphatic vessels. Immune cells use these two networks to patrol the body, so that they are ready to attack if a pathogen enters and to protect against derailed (cancer) cells.
Just as an army can have an army and an air force, the immune system can be roughly divided into two major parts. These are the innate and the learned (or acquired) immune system.
The innate immune system is always ready, but has no memory
The innate immune system is always ready immediately and works quickly. It is also called the non-specific immune system, because it reacts to all invaders and not specifically to a particular pathogen. This part has no memory. For the innate immune system, there is no difference in the immune response when someone comes into contact with the same pathogen for the second or third time.
The innate immune system consists of all kinds of immune proteins and different types of immune or white blood cells. Important cells of the innate immune system are monocytes, macrophages, granulocytes, 'natural killer' cells and dendritic cells. They all have different functions, which sometimes overlap somewhat. Granulocytes are very good at fighting bacterial and fungal infections. Virus infections mainly involve macrophages, 'natural killer' cells and dendritic cells.
Macrophages are in all tissues and are, as it were, on guard. Macrophages can be described as 'big eaters', as they effectively eat and break down bacteria, fungi and viruses. This happens through a process called phagocytosis. Macrophages (and most granulocytes) are therefore among the phagocytic immune cells. Macrophages can also pass on information to other immune cells. They do this by releasing signal substances, like lighthouses, that show other immune cells the right way.
'Natural killer' cells recognize cells that are infected with a virus and then give instructions to this cell to destroy itself. Because a virus depends on a host cell to replicate itself, it is beneficial if the host cell destroys itself, because this also destroys the virus.
Dendritic cells resemble macrophages and are also on guard in the tissues. But their main function is not to eat many pathogens, but to taste them, so to speak. They can then go via the highways in our body to the lymph nodes and the information
to the cells of the learned immune system (the lymphocytes). They can therefore be seen as messengers and conductors of the immune system because they provide and direct the lymphocytes with the right information.
The learned immune system is very specific and has memory
In contrast to the innate immune system, the learned immune system is very specific and develops memory cells. It is slower and takes about 7-10 days to develop properly when someone comes into contact with a pathogen for the first time. But after that, the protection against the same pathogen is long-term and in some cases even lifelong. This does not mean that someone can no longer get sick from this specific germ, but if the learned immune system is reactivated, the protective effect is faster and someone will become less ill than the first time. A person does not have this part of the immune system at birth, because one must first have been in contact with a pathogen in order to be able to produce memory cells. This has to be redeveloped for each new pathogen. The learned immune system is therefore built up throughout life.
Cells of the learned or adaptive immune system are called lymphocytes. Lymphocytes respond very specifically because they can recognize intruders with the help of certain recognition molecules on their surface. These recognition molecules, or receptors, recognize a small piece of the intruder very precisely. This can also be compared to a key-lock reaction. The lock will only open if the correct key fits. For example, a lymphocyte that has a receptor for a molecule of a flu virus will not respond to a Salmonella infection and vice versa. Lymphocytes can develop into a memory cell. Once they have seen a particular intruder, they react more quickly if it tries to enter the body again. This has the great advantage that a person who has had a certain disease does not become ill or becomes less ill the second time.
There are two types of lymphocytes called T cells and B cells. T cells can be further divided into killer T cells and helper T cells. Killer T cells are
the snipers of the immune system. They can recognize virus-infected and derailed (cancerous) cells and then give very specific instructions to the cell to destroy itself. In terms of function, they resemble the 'natural killer' cells. The helper T cells can alert and activate other immune cells if necessary. When a B cell is activated, it secretes antibodies. The B cell becomes a munitions factory, as it were.
Antibodies are also called antibodies or immune globulins. Antibodies have two different sides. One side (the 'head') is very specifically directed against a molecule (antigen) that is on a pathogen. The other side of the antibody (the 'tail') binds to a group of recognition molecules that occur on immune cells, such as macrophages. By binding with the 'head' to the antigen on the germ and via the 'tail' to the recognition molecules on the immune cells, antibodies form a bridge, as it were. This activates the immune cell and clears the germ.
Antibodies are in breast milk to protect a newborn baby from germs. Nowadays, antibodies can also be isolated from the blood of healthy volunteers, after which they are given as a treatment to patients who do not make enough antibodies themselves. This is called intravenous immune globulins (IVIG). Finally, antibodies are now also made in the laboratory as a treatment against infections and cancer.
Interferon plays an important role in viral infections
Shortly after an infection, a flood of reactions occurs. In a viral infection, the production of the signaling molecule interferon plays a major role. This is produced by cells that have been infected by the virus. Interferon does two important things. On the one hand, it interferes (interferes) with the multiplication of the virus. The name interferon is derived from that. On the other hand, interferon activates certain cells of the immune system such as the macrophages and the 'natural killer' cells that attack the pathogens. The dendritic cells also do their bit. They 'taste' the infection and then migrate to the lymph nodes where they pass the information on to the lymphocytes.
Some pathogens secrete certain toxic substances (toxins) that induce fever. Febrile substances can also be secreted by immune cells in response to the infection. Fever helps to fight the infection because it prevents the growth of certain pathogens. It also makes our immune system work harder. Unfortunately, fever can also make someone feel very ill. If the disease lasts longer than a few days, the learned immune system comes into action. The lymphocytes in the lymph nodes have received information from the dendritic cells and start to multiply to fight the pathogens. This causes the lymph nodes to swell, which is often noticeable in the case of a sore throat, for example.
The SARS-CoV-2 virus is new to humans. That is why virtually no one has built up resistance yet. Only people who have been exposed to the SARS-CoV-2 virus in recent months and have survived the infection are likely to have built up a protective immune response. But all other people are unprotected. That is why the virus can spread so quickly and why the pandemic emerged. This is a big difference between, for example, SARS-CoV-2 and flu (Influenza).
The immune system of different patients responds differently to the SARS-CoV-2 virus
The course of the disease varies from patient to patient. For most (estimated 80-85%) people, it feels like a cold or mild flu, while other people (15-20%) develop severe pneumonia and spend weeks on a ventilator or die. At present, too little is known about the virus to understand exactly why one person becomes ill and another does not. But what is similar to other types of infections is that the immune system response is very different in different patients. This probably plays a major role in the course of the disease.
A normal immune response is very balanced. The Kedzierska Lab in Melbourne has investigated the immune response and immune cells of a patient with a mild SARS-CoV-2 infection. In this patient, various parts of the immune system were quickly activated, as is also seen in other infections. With a mild infection
the immune system therefore appears to respond normally, causing the sick person to improve. It is not yet completely clear how the immune system responds in seriously ill people. It appears that the immune system in these patients reacts too strongly and goes into overdrive. In those cases, far too many signaling substances are produced, which cause a lot of damage to tissues and cause inflammation in the lungs. This is also called a cytokine storm and could explain why the condition can worsen over a short period of time in some patients. In this case, a serious inflammatory reaction occurs, causing the mucous membrane in the alveoli to thicken. This makes it more difficult to absorb oxygen and get it into the bloodstream. The lungs also fill with fluid and pus (= dead immune cells) and breathing becomes increasingly difficult. It is still unknown why the immune system suddenly runs wild, but it is more often seen in response to a respiratory pathogen. With the Spanish flu, for example, this was also the case in seriously ill patients.
Women often have better immune systems
Women seem to respond better to the infection than men. On average, two out of three patients with severe symptoms are men. Women generally have better defenses. This is probably because the regulation of the immune response is controlled, among other things, by the X chromosome. And because women have two X chromosomes and men only one, women's immune systems are better regulated than men's. Excess weight appears to play a negative role. The underlying mechanisms are still unknown, but several studies have shown a relationship between obesity and impaired functioning of the immune system. When children become infected with the SARS-CoV-2 virus, the chance of them becoming ill is surprisingly small. They usually experience either no symptoms or very mild symptoms. Few children end up in intensive care, although there are exceptions. Yet it is clear that children respond better to the infection than adults. Children may have more innate immune cells that are involved in fighting a SARS-CoV-2 infection. However, it may also be the case that children have fewer attachment molecules to intestinal and lung cells.
Group protection
An important reason why the SARS-Cov-2 virus is so dangerous is that it is new to humans. As a result, virtually no one has built up resistance (= immunity) yet. Everyone is susceptible to infection with this virus, which allows it to spread unhindered and cause many victims.
Group protection or herd immunity protects the vulnerable people in society
Group protection, or herd immunity, can protect vulnerable people in a society against serious diseases, such as SARS-CoV-2. When many people in a community are immune to a particular pathogen, the spread is quickly stopped. Although a non-immune person can become ill himself, he cannot infect immune persons in the environment and the disease will fade away. This significantly reduces the risk of infection for vulnerable people.
How many people in society must be immune for effective group protection depends on the multiplication number (R). More people should be immune to a highly contagious pathogen than to a less contagious pathogen. Measles, for example, is a highly contagious disease. One sick person can infect up to 18 other people (R0=18). If no one were yet immune, measles could spread alarmingly quickly (from 18 people to 364 to 5,832 to 104,976 and so on).
Using the R, it can be calculated how many people must be immune to have good group protection. As a rule of thumb, an epidemic cannot occur if the percentage of immune people is higher than ((R0-1)/R0) x 100%. For measles that is ((18-1)/18) x 100% = about 94%. Group protection is jeopardized if that percentage becomes lower. It is therefore worrying that measles vaccination rates have declined in recent years. For SARS-CoV-2 with an estimated R0 of 2.2, ((2.2-1)/2.2) x 100% = approximately 55% of people need to be immune to stop the pandemic. A slightly wider margin is used and it is therefore assumed that group protection occurs when 60% of the people are immune. If it is possible to reduce the number of infections (the multiplication rate decreases) by taking measures, fewer people with resistance are needed to achieve this.
Each country takes measures depending on the situation in that country
Countries around the world are taking measures to stop the pandemic. There are different scenarios for this. Scenario 1 is doing nothing to obtain group protection as quickly as possible. This approach has the advantage that everyone is exposed and the 60% of people with resistance are reached most quickly. However, this would be accompanied by a great loss of life. The United Kingdom, for example, initially planned to choose this scenario, until it became clear that this would mean that almost 40 million people would become ill in a short time, of which 8 million seriously. The health care system, and especially intensive care units, would become completely overwhelmed with patients and this would lead to a healthcare infarction that would cause hundreds of thousands of patients to die.
Scenario 2 does exactly the opposite by imposing a so-called lock-down on the country and banning people from leaving their homes. The advantage of this approach is that the number of infections is limited as much as possible in the short term. However, a risk is that time is, as it were, frozen and no group protection is built up at all. Once the lock-down is lifted, the situation has not changed and only one infected person needs to enter the country to start all over again.
Most countries choose a middle path. This is based on models that calculate the expected number of hospital admissions and intensive care admissions in that country. It will then be determined which measures will ensure that the maximum capacity of the hospitals is not exceeded. This means that a country with few infections may need to take fewer measures than a country with many infections. It may also be the case that measures need to be tightened if the number of infections increases in a country with few infections until then. This policy is usually referred to as “flatten the curve” and must ensure that the predicted wave of infections remains below the limit of the maximum possible hospital admissions. Italy, France and Spain, for example, have opted for a lock-down, while Sweden has opted for an approach closer to scenario 1, because there are still few infections. Universities are closed and large public events are prohibited, but catering, schools, shops and gyms are still open. No one knows at this point what the best approach is. That too will only become clear afterwards.
In the Netherlands, an 'intelligent lock-down' has been opted for and citizens are asked to take their own responsibility. People should work from home as much as possible, go out as little as possible if not necessary and not visit each other with more than 3 people. Schools are closed and there is a group ban for more than 2 people. The aim of this policy is not necessarily to achieve herd immunity, but to spread the number of infections over time. This will hopefully not everyone get sick at the same time, so that the ICU departments do not become overloaded. An additional advantage is that group immunity will slowly develop. Naturally, the situation is being closely monitored and measures will be tightened if either the number of infections does not fall quickly enough or if people do not sufficiently follow the prescribed measures.
It is extremely difficult to predict how long it will take before herd immunity is achieved. Currently, based on current figures and models, it is assumed that approximately 60% of people must have been infected and must have resistance
The Netherlands has opted for an 'intelligent lockdown'
built up. The problem is that it is still unknown how many people have been infected with SARS-CoV-2. There are indications that there are people who have been infected, but hardly have any symptoms. It would be good news if this involved many people, because group protection would then be achieved more quickly. However, if this is only a few people, this is not going to help. It is also not yet clear how long people will remain immune against SARS-CoV-2. Milder variants of the coronavirus, which only cause the common cold, are known to only protect people for about a year. It is unknown whether this is the same with the more aggressive SARS-CoV-2 variant. If people are indeed susceptible again after a year, it will take longer before herd immunity is achieved. The availability of a vaccine (see page 22) would greatly accelerate the achievement of herd immunity.
Testing everyone who has cold symptoms would provide a better picture of the number of people infected with the SARS-CoV-2 virus. Although people without symptoms would still be missed, it would still provide a better picture of the expected group protection. However, there are two problems with this approach.
Testing everyone leads to major shortages and a false sense of safety
The first problem is that in this day and age, in addition to SARS-CoV-2, flu and cold viruses are also prevalent. There are a lot of people with cold complaints and fortunately the chance that someone is actually infected with the SARS-CoV-2 virus is still small. Testing all these people would lead to major shortages. There is a scarcity of tests and therefore mainly healthcare staff, patients admitted to hospital and vulnerable patients outside hospitals are tested. To ensure that enough tests remain available for these risk groups, people with mild complaints are not eligible. These people must recover their illness at home and go into self-quarantine, together with their family members or housemates.
The second problem is that no test is error-free. A percentage of patients will incorrectly receive the result that no disease is present. This is a false negative score. At the same time, healthy people will incorrectly see the rash
get 'disease' (false positive score). The numbers of false negative and false positive scores determine the reliability of a test. On the one hand, it is now clear that patients can receive a false negative score, while they are tested positive two days later. During this time, people are wrongly reassured and do not take proper precautions to avoid infecting others. On the other hand, mass testing of people with cold complaints will lead to many false positive results, which will cause a lot of unnecessary anxiety. That is why in the Netherlands, targeted testing is mainly carried out when there is already a serious suspicion of infection with the SARS-CoV-2 virus. The RIVM and Sanquin will soon start with a large sample to gain a better insight into how the SARS-CoV-2 virus has spread across the Netherlands.
Antibody titer determination provides information about the development of group protection
People who have been infected with the SARS-Cov-2 virus produce antibodies, even when the symptoms have gone unnoticed. The number of antibodies can be determined in the blood and is called a titer determination. This test does not determine whether someone is infected at that moment, but whether someone has already come into contact with the virus. Initial results suggest that the antibody titer is low in people with mild infection. By measuring the antibody titer in large groups of people, one can find out how many people in society have become infected with the virus, have developed immunity and how group protection is developing. The problems mentioned are also the same in this test. It is understandable that everyone wants to know as soon as possible whether they have been infected. But this will lead to major shortages of testing. In addition, this test will also have false negative and false positive results. It is therefore important that the tests are carried out in a coordinated manner.
The RIVM and Sanquin (the blood bank) will collect blood samples from people from all over the country and from all age groups. The antibody titer will be determined at several times over a number of months. By testing the same people multiple times, the results become more reliable. Moreover, in this way it can be investigated how long antibodies remain present and whether they are of good quality. In addition, it is mapped out how group protection develops over time in the Netherlands.
Vaccination.
The best way to get group protection safely is vaccination. It is therefore important that a good vaccine against SARS-CoV-2 is quickly available.
A vaccine is vital
What is the principle behind vaccination? Normally the immune system reacts faster and stronger when it comes into contact with the same pathogen for the second time, because it has built up memory. This means that people either don't get sick at all the second time or get better faster. This can be imitated with vaccination. The first exposure then occurs by giving a weakened or harmless form of the pathogen. The immune system will respond to this as if there were a real infection and immunological memory is built up, without people actually becoming ill. When someone then comes into contact with the real pathogen (in this case SARS-CoV-2), someone does not become ill or becomes less ill.
Of course, this does not only apply to SARS-CoV-2. Thanks to the National Vaccination Program, children are protected as best as possible against dangerous diseases. And this protects everyone, including the vulnerable people in society. It is therefore alarming that more and more people think that vaccination is unnecessary or even dangerous. This reduces the vaccination rate against serious diseases and reduces group protection threatened. The Corona crisis emphasizes the importance of vaccinations, which are generally safe and effective.
Vaccines are medicines. That is why they must meet very strict requirements before they can be used. This is monitored by the Medicines Evaluation Board (MEB) and the European Medicines Agency (EMA). The safety and effectiveness of vaccines are continuously monitored. This keeps track of how often and what side effects there are. Side effects of vaccination are usually mild. The vaccination site may become red, swell and hurt. Short-term headaches, fatigue and mild fever are also relatively common. These symptoms are due to the activation of the immune system. Very occasionally, the immune system can overreact and an anaphylactic shock (very serious allergic reaction) can occur. However, the risk of serious side effects from a vaccination is many times smaller than from the infectious disease.
There are currently many different initiatives to develop a vaccine. Why not collaborate to have one vaccine ready as quickly as possible? Of course, this also has to do with company interests, but it is also good to try out different things. SARS-CoV-2 is new and it is not yet clear how the immune system responds to it. No one knows yet what the best way to develop a vaccine is. It is of course important that the information learned is made known, so that other scientists can learn from it.
The immune system appears to respond normally to SARS-CoV-2
SARS-CoV-2 does not appear much to mutate
In any case, the first reports seem favorable. Research by the Kedzierska Lab in Melbourne with a first COVID-19 patient has shown that a normal immune response is generated in the case of a mild infection. This suggests that vaccination could also induce an immune response. In addition, SARS-CoV-2 appears to mutate little, according to Peter Thielen, a geneticist at Johns Hopkins University in Baltimore. That is also very important for the development of a good vaccine. Because vaccines are very specific, they do not protect well against a pathogen that has changed. This is a major problem with flu vaccines, for example, because flu viruses continuously mutate and therefore change. That's why we have to new flu vaccines are developed every year. In the case of SARS-CoV-2, hopefully only one vaccine is needed, because the virus is less changeable.
Three different forms of a vaccine
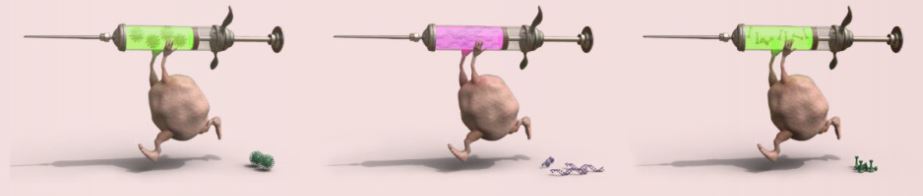
Left: a classic vaccine with weakened or dead pathogens. Middle: Moderna Therapeutics' vaccine with only pieces of RNA. Right: the vaccine with isolated protuberances being developed in Wageningen.
From 1e volunteers have been vaccinated
The first volunteers are now being vaccinated. This happens in a new way. In the past, most vaccines contained a live, but weakened pathogen. This causes the immune system to respond well, preventing healthy people from becoming infected. However, this type of vaccines can cause problems in people whose immune system does not work properly or is weakened. To increase safety, inactivated, dead vaccines are mainly used nowadays. These consist of pieces of pathogens that cannot multiply. This new vaccine, developed by the company Moderna Therapeutics, goes one step further. There is no virus at all, but only a piece of the genetic code of the virus is injected (in this virus it is RNA). This piece of RNA codes for the virus spikes that SARS-CoV-2 uses to enter the human cell. This causes the body to only produce these protrusions, but not the rest of the virus. And hopefully the immune system will also be activated, causing protective antibodies to be produced against the protrusions. Whether this works will become clear in the coming months.
The University of Wageningen has a slightly different approach. These researchers have developed a technique in which insect cells produce large amounts of protrusions. These can then be isolated and used as a vaccine. The company Janssen Vaccines Leiden has a different approach, involving use is made from a harmless cold virus, in which a small piece of RNA from the SARS-CoV-2 virus has been inserted. This vaccine is expected to be tested in humans for the first time in September 2020. The safety and effectiveness of these vaccines must be extensively tested before they can be widely used. It is expected to take at least another year before a vaccine is available.
Because the development of a vaccine will likely take some time as the crisis rages, other things are also being tried to combat the disease. For example, the Radboud University Medical Center is investigating whether a vaccine against tuberculosis strengthens the immune system. This vaccine will not induce a specific immune response against the SARS-CoV-2 virus, because it is directed against Mycobacterium, which causes tuberculosis. But this vaccine contains a weakened live bacteria that remains present for several months after injection under the skin. This activates the innate immune system. Previous studies have shown that this strengthens resistance to, among other things, the flu and malaria. It is now being investigated whether this also helps with a SARS-CoV-2 infection.
Healing antibodies
During a recent Ebola outbreak, it was discovered that antibodies from people who had recovered could be used to treat patients. It is now being investigated whether this is also the case with a SARS-CoV-2 infection. That is why Sanquin has started collecting blood plasma from recovered corona patients. This is done in close collaboration with hospitals, researchers from within and outside Sanquin, the GGD and the RIVM. The plasma can then be given to seriously ill patients in the hope that it will stop the spread of the virus in the body and alleviate symptoms. This may even prevent patients from having to be admitted to intensive care. The first study has currently started at the Erasmus Medical Center. It is also being investigated whether this plasma can be used to protect healthy people who come into contact with patients against infection.
Sanquin will also try to turn this into a medicine in the future, but at the moment it is not yet a generally available treatment for patients with COVID-19. For the time being, plasma will still be scarce until enough patients have been cured.
Antibodies prevent the SARS-Cov-2 virus from infecting the host cell.
Another disadvantage of this treatment is that there is only temporary protection. No immunological memory is built up and the antibodies given will gradually disappear from the body. But this treatment could be a solution for people in healthcare, vulnerable people and the elderly.
Nowadays, antibodies can also be made in a laboratory. Scientists from Utrecht University and the Erasmus Medical Center have found an antibody that could inhibit infection with SARS-CoV-2. This is now being produced on a large scale and will also be tested in a clinical trial.
Experimental virus inhibitors
It will take some time before vaccines and antibodies are widely available. That is why alternatives are being sought that slow down the virus in COVID-19 patients. There are a number of medicines that have proven effectiveness in other infections. The drug Remdesivir has been tested in Ebola patients and ensures that the Ebola virus can no longer multiply. It is now being investigated whether this medicine can also inhibit the multiplication of the SARS-CoV-2 virus. A second drug is the malaria drug chloroquine, because it is thought that this drug could inhibit the entry of the SARS-CoV-2 virus into the cell. Treating COVID-19 with these drugs is still experimental in both cases. Because these drugs can also have serious side effects, it must first be thoroughly investigated whether patients actually benefit from this treatment before it can be administered on a large scale.
Cytokine storm inhibitors
Another drug under investigation is called camostat. This drug does not attack the virus, but an attachment molecule on the cell. If this drug prevents the virus from binding and entering, it also inhibits the infection. After all, a virus depends on a host cell for multiplication. It is also being investigated whether it is possible to inhibit the worst symptoms. The abundance of signaling molecules, the so-called cytokine storm, is probably the cause of the severe pneumonia in a number of patients. There are already medicines on the market that can inhibit some of these signaling substances. It is now being investigated whether this also works for COVID-19.
Finally: Important points
- The SARS-CoV-2 virus is new to humans. That is why not everything is known yet and information can change due to advancing insight.
- Symptoms of the infection are highly variable. Even people without symptoms can be contagious. Therefore, please adhere to the regulations. Stay at home as much as possible, keep your distance and wash your hands often with soap.
- The measures taken will reduce the infection rate R. By lifting the measures too early, infections could increase quickly again. So it is important to persevere.
- Group protection protects the vulnerable people in society. Vaccination accelerates the achievement of group protection.
- Pay attention to each other.
Dutch Society for Immunology (NVVI)
Text:
Prof.dr. Marjolein van Egmond, Amsterdam UMC
Illustrations: Prof.dr. Georg Kraal, Amsterdam UMC
Prof.dr. Marjolein van Egmond, Amsterdam UMC Prof.dr. Marieke van Ham, Sanquin
Prof.dr. Georg Kraal, Amsterdam UMC Prof.dr. Reina Mebius, Amsterdam UMC Prof.dr. Annemiek van Spriel, Radboud UMC Dr. Edward Knol, UMC Utrecht
Annelot Breedveld MSc, Amsterdam UMC Saskia ter Braak, UMC Utrecht
Stella from de Bosch, Amsterdam UMC Mariska van der Zee, Amsterdam UMC
https://www.dutchsocietyimmunology.nl/