Looking back on the introduction of the proven "Safe and Effective" MRNA vaccines, the "effective" aspect has disappointed: vaccination figures under COVID oversen are not reassuring and are at most unintentionally released. 80% of the deaths to COVID-19 during the Deltagolf was vaccinated1See steig.nl: The big secret of the Deltagolf. More than 90% of the participants in the Long-Covid government project have been vaccinated2See What disease is the post-Covid report?, ook iets dat zou niet moeten kunnen na een trial die geen twijfel overliet aan effectiviteit. Maar het klinkt ook niet dat je zegt: “veilig” want als het niet effectief is tegen Covid, wat doet het dan wel? Het is immers geen zoutwateroplossing en de gestegen sterfte aan alle oorzaken en vele meldingen van bijwerkingen wekken ook geen vertrouwen. Dat zou echt niet zo moeten zijn na een trial die het sein “veilig” had opgeleverd. Pfizer heeft kennelijk een andere definitie van "veilig".
The basis of "safe" is becoming smaller
In the public discussion, it is assumed that the Pfizer trial3The Pfizer report: Summary Basis for Regulatory Action - COMIRNATY With over 43,000 participants provided sufficient evidence for the designation safe & effective.
Everyone who has viewed critical vaccinations studies has probably thought: "If a sample of 43,000 participants offers sufficient safety to inject the entire world population, why have mega studies done in the past with hundreds of thousands or even millions of test subjects?" The statistical knowledge that such an research design required existed in the 1950s and 1960s. Yet those investigations were not a little bigger: they were 40 times as large! And for some time we can finish it with a few tens of thousands. Why?
The larger, the more precise. The smaller, the more statistical uncertainty, which increases the possible insecurity.
Although 43,448 people were enrolled, the primary safety analysis on which the “safe” claim is based involved fewer participants because not everyone completed a full follow-up. The safety analysis involved 37,706 participants, which again makes you wonder how those 5,700 dropouts were selected. In any case, that results in an upper limit of ±10,000 additional deaths per 12 million vaccinated within 2 months. More precise is not possible. But a median follow-up duration of two months is also reported and this means that an unknown part of the group was followed for less than two months4“Safety was evaluated in 37,706 participants who received at least one dose of vaccine or placebo; 18,860 received BNT162b2 and 18,846 received placebo. At the time of the data cutoff, a median of two months of safety data was available for 18,860 vaccine recipients and 18,846 placebo recipients.”
— Polac Fp, et al. No, 2020. Dat levert een hogere bovengrens op die we niet precies kunnen berekenen omdat we de grootte van het uiteindelijke sample niet kennen. Dat zal dus kleiner zijn, en hoe kleiner het sample, hoe groter de onzekerheid. Daardoor gaat de statistische bovengrens verder omhoog en wordt "veilig" steeds minder veilig. Ik zal de grootte aangeven met <38.000.
The cut-off date chosen by Pfizer (November 14, 2020) determined how many participants had completed the two months. This therefore depends on the registration date of each participant and accurate registration thereof. Because individual data is not public, data integrity is only guaranteed by the manufacturer, who must be alert to possible 'sloppiness'. The fact that it is difficult to report imperfections is evident from how reports from an employee of the Pfizer trial, Brook Jackson, were handled.5 Interviews met Brook Jackson, die later in The BMJ (2021) the disorderly data collection and defective monitoring described)6Brook Jackson met video in The BMJ: https://www.bmj.com/content/375/bmj.n2635.
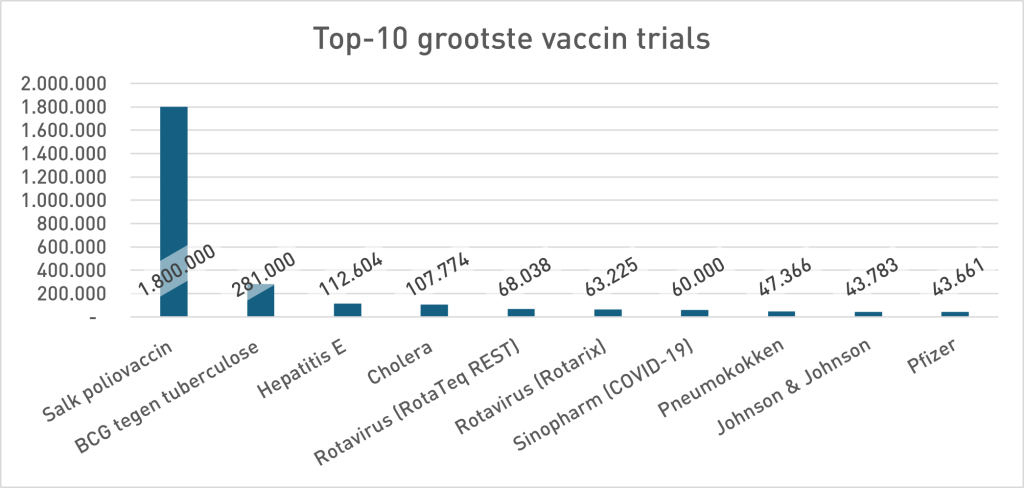
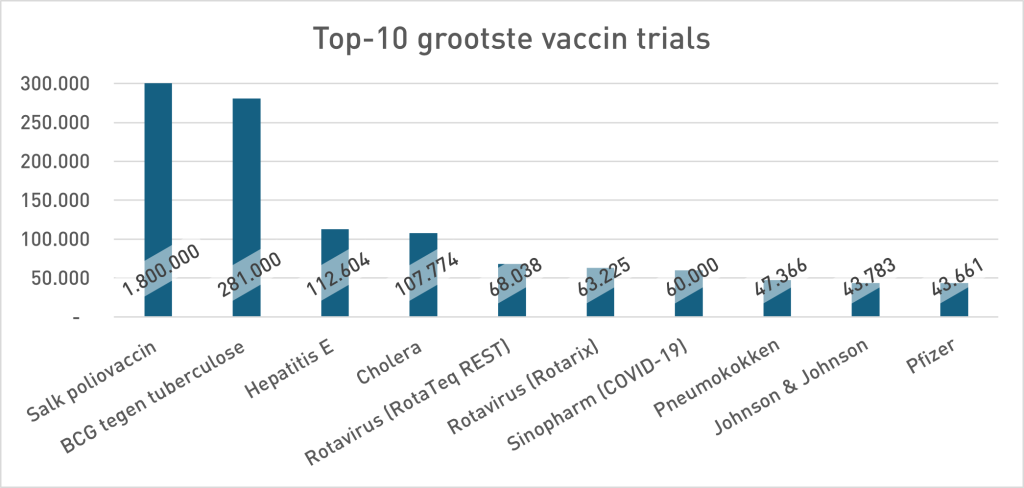
In the phase 3 security study of Pfizer (Sample <38,000), 21 people died in the Vaccing Group and 17 in the placebo group. 23% more death in the vaccinated group. Pfizer's conclusion was: no statistically significant difference in death, so the vaccine is "safe". For the argumentation theorists among us it is immediately as ready as a lump: absence of evidence is not proof of absence. Not only a statistical rule but a classic fallacy: the non sequuitur7Uitleg does not follow by chatgpt. But pharmaceutical companies don't think that way, so let's look in their own history to see whether that is a new insight. The study design (size) at Pfizer contrasts sharply with previous vaccine studies (move your mouse over the graph to zoom in):
If Pfizer can manage with approximately 44,000 participants (and < 38,000 in terms of safety), why were such numbers earlier than necessary? This could be related to a change in the meaning of the concept of 'safety'.
Waar denken we dat "veilig" voor staat?
Als je iemand met een visuele beperking helpt met "Het is veilig, u kunt oversteken" dan zeg je dat niet als je denkt dat er een kleine kans is dat hij wordt overreden. Dat zeg je pas als je zeker weet dat hij veilig de overkant zal bereiken. Als burger vaar je blind op farmaceuten. Maar die hebben andere standaarden.
In clinical practice, “safe” usually means: there is no significant difference from placebo in this study population, in the limited period of the trial. So that's the fallacy. In everyday speech, “safe” means something completely different, namely: no real risk of death or serious damage. But again it is unclear what “none real risk ”. That is not"no Risk ”. In which numbers should we think? Let's assume that a risk of 1 extra death per 10,000 punctures would still be considered acceptable (we will come back on risk categories later). It is the second safest category according to the official EMA/WHO classification. (Often:> 1/10, sometimes: 1/1,000-1/1,000, rarely: rarely: rarely: rarely: rarely: rarely: rarely: rarely: rarely: reld the ownership: 1,000, reld the ownership: reld the ownership: reld the ownership: reld the ownership: reld the ownership: reld the owner: reld the ownership: rarely: rarely: 1,000: rarely: 1,000: 1,000 and 1,000! 1/10,000-1/100,000.)
How big should a trial be?
We assume a hypothetical maximum risk: the additional chance of dying within two months (the duration of the trial!) after vaccination may not exceed 0.01% (1 in 10,000). For a population of 12 million vaccinated people (NL), this means a maximum of 1,200 deaths in two months - which you can also question, but we agreed to accept this as being 'safe'. It should also be weighed against the severity of the disease. What is also missing is the age aspect. All ages are tarred with the same brush, which raises ethical questions and can lead to statistical distortions.
Om uit te sluiten dat het risico groter is dan 0,01%,, zijn meer dan 1 miljoen proefpersonen nodig. (zie kopje Veldwerk, onderaan). Wil je daarbij een likelihood ratio van 100% bereiken dan worden het vele, vele miljoenen. Dat heeft dus geen zin meer als 'trial', dat is "uitrollen en zien wat er gebeurt".
However, Pfizer worked with < 38,000 test subjects. This means that you cannot statistically notice a small difference such as 1:10,000. In fact, with such small numbers you can only rule out that no more than approximately 10,000 to 12,000 deaths would occur in the first two months among 12 million vaccinated people.
This means that the Pfizer safety trial indicates that the total mortality in the population can remain the same by half due to the vaccinations, because that is what the numbers come down to. Everything in between is therefore declared acceptable (because 'safe').
Moreover, it is important that this calculation only applies to the duration of the trial: two months. We now know that side effects and deaths sometimes only become manifesto after 3–4 months (± 100 days) and possibly later. But also the suspected height of that risk of death would not have been observed in this underpowered study8Observational estimates appear to indicate a risk of 0.02% for the baseline series. That would be 2400 deaths for two months. That falls well within the insensitivity range of this design..
The ethical dimension
Medications are given to sick people. A small extra risk can be acceptable there, because the patient runs a high risk without treatment.
Vaccines, on the other hand, are given to healthy people, to children. That makes the balance fundamentally different. Can you let a healthy (younger) individual risk of death to prevent deaths elsewhere (in the elderly)? That is not a purely scientific, but above all an ethical and possibly culture -related question. And that is why perhaps a political one for a referendum.
"Mogen burgers gevaarlijke injecties nemen vanwege angsten, wat ook wel 'informed consent' wordt genoemd?"
Age dependence
Like many other disorders, COVID mortality is strong age-related. The percentage aboutmortality per age group is comparable across all age groups. The trials did not even remotely have the power to prove safety and effectiveness per age group separately. Results for an “average subject” were simply generalized to all ages. That might still be justifiable if the goal had been herd immunity. It goes without saying that the whole argument of herd immunity did not apply to an injection anyway. An intervention that did not reduce infections and was usually administered at the wrong time because it was effective for such a short time (what a panic football, if you put it all together again.)
Semantic and framing
The words themselves have suddenly evolved:
- "Vaccine" stood for an innocent first encounter with a pathogen, so that the body builds up immunity without any risk. With mRNA and vector vaccines, it has become a medicinal intervention because immunity is so temporary that the drug should preferably be administered a few weeks before infection - an impossible timing, as the 'boosters' have proven. The distribution through the body is also very different. Traditionally, the further away from the injection site, the weaker. mRNA vaccines are micro-submarines that can travel anywhere in the body and only release their active cargo upon arrival in a cell.
- “Trial” once stood for long-term, large-scale field studies (polio, BCG, hepatitis E, cholera). The Covid-19 trials were relatively small and short. Looking at the required power and the research carried out, they were mainly for the stage, shortcuts to approval. Meaningless data was interpreted as safe. The absence of evidence for excess mortality - the predictable outcome in these small samples - was framed as evidence to the contrary. This invites you to look at other vaccine studies because as we can see from the top 10 graph, almost all studies are numerically below the Pfizer trial discussed here. They only reach 11th place and much lower.
Conclusion
- Pfizer’s Trial did not proved that the vaccine safe was in the everyday sense of that word. It only proved that there was no significant difference in mortality over a limited period of time, compared to placebo. But absence of evidence is not evidence of absence.
- It was known in advance that this trial would not even remotely demonstrate that the safety of the vaccine would be at stake. The popularity of this range of sample sizes is understandable, from a pharmaceutical perspective. A lot has to happen if this is to produce a hard (significant) safety signal.
- To demonstrate true safety, much larger trials are needed — and that has been done historically. That's how it started. The claim “safe and effective” has become the result of framing and repetition, not of hard science.
- De cijfers zijn een harde grens: minder dan 12.000 doden in twee maanden na de prik, daar is de trial op gedimensioneerd. Dat is de uitspraak “veilig” ook. Minder dan 12.000 overlijdens in twee maanden is "veilig". Het zijn waarschijnlijk niet meer dan 2.000 vaccindoden per 2 maanden. Dat valt ruim binnen de marge. Tweeduizend.
Fieldwork at Chatgpt and Grok
Question
Suppose we want to demonstrate that the extra risk of death in a certain medicine must be smaller than 0.01%. We are going to use a placebo group and see that the background mortality is 0.1%. How many participants do I need in 2 months to draw a conclusion with 95% certainty about the safety of the medicine?
https://chatgpt.com/c/68a5c856-3ecc-832f-9a52-3be4304100cf
Chatgpt Conclusion: Over with 95% certainty to conclude that it extra death risk < 0,01% is with a background of 0,1% in 2 months, do you-in a solid RCT setup—about 1–2 million participants per arm Needed (depending on the desired power) ..
And then we accept still That 1200 people could die from the medicine. That can ultimately be (much) less, but we don't know that. The more certainty we want, the more participants we need.
Same question for grok
https://x.com/i/grok/share/ssrWdWIyuYkoMpFpJmJzSBfbT
Answer
941,312 participants, is the outcome of the calculation. The minimum remains in the order of the 1 million participants.
Question:
Suppose the willing to demonstrate that a medicine is safe and we use a placebo and test group with both 13,000 people. In 2 months, 21 die in the test group and 17 in the placebo. What statement can we make about the safety of the drug?
And the follow -up question:
Calculate upper limit of the extra chance of dying from the medicine
https://chatgpt.com/c/689f53f8-4850-8323-ae9a-1745ab019df5
The answer to the first question is:
- There is no statistically significant difference in death between medicine and placebo.
- So we cannot prove that the drug is more unsafe, but also not prove that it is safe-that requires more data or longer follow-up.
- In statistics, "absence of evidence is not proof of absence".
A clear answer! But what is the maximum death of death because of the medicine, so in this case the mrna vaccine. Also a clear answer there:
0,1087% absolute extra kans.
In other words, Pfizer guaranteed us through this trial that the number of deaths in the first 2 months will be less that 12,000. Of course that is an upper limit, the chance is small that it will be more than this. Pfizer's claim that this study has shown that the MRNA vaccine is safe can only be drawn if you accept that 12,000 deaths are safe enough in the first 2 months.
In summary:
- In order to rule out with 95% certainty that more than 1200 people die in the Netherlands in the first 2 months after vaccination, you have to take around 1 million people in your trial.
- If you only take 19,000, you can only promise that no more than 12,000 people will die in the 2 months after vaccination. Each value between 0 and 12,000 is eligible.
- The conclusion that 21 deaths after vaccination died against 17 unvaccinated has no relevance with regard to the statement that vaccination is safe.
Chatgpt's powerlessness
For the enthusiast: Self -reflection of Chatgpt who explains why she spreads obvious disinformation without any problems.
https://chatgpt.com/share/68a01e03-b538-8007-a657-0af79b6f8f6a
Footnotes
- 1
- 2
- 3The Pfizer report: Summary Basis for Regulatory Action - COMIRNATY
- 4“Safety was evaluated in 37,706 participants who received at least one dose of vaccine or placebo; 18,860 received BNT162b2 and 18,846 received placebo. At the time of the data cutoff, a median of two months of safety data was available for 18,860 vaccine recipients and 18,846 placebo recipients.”
— Polac Fp, et al. No, 2020 - 5
- 6Brook Jackson met video in The BMJ: https://www.bmj.com/content/375/bmj.n2635
- 7Uitleg does not follow by chatgpt
- 8Observational estimates appear to indicate a risk of 0.02% for the baseline series. That would be 2400 deaths for two months. That falls well within the insensitivity range of this design.

Nice article, men!
The Pfizer trial with the about 40,000 people was performed with an MRNA product that was synthesized according to process 1.
The product that was brought to the market with provisional approval was prepared according to process 2. This was only tested on a few hundred people.
It was known that according to process 2, the product 2 contains impurities than the first product. Furthermore, the consistency of the qualitir of product 2 was not in order, certainly in the beginning. The EMA knew that and noted that at the points for improvement. Later it was also found that there was batch to batch variation and "Killerbatches" were demonstrated.
Effective and safe. A new technique on which almost all available Covid "vaccines" were based. For me, that remains the most incomprehensible, how is it possible to choose worldwide for mrna and that all available "vaccines" became available in such a short period relative to each other, versus the old -fashioned method?
Because this mrna technology has been so "safe and effective", more and more mrna "vaccines" are being developed, for various diseases and other all caus mortality. With priority for bird flu, that will be the next planning. I suspect that the research groups for those "vaccines" will be even smaller, so even safer and more effective!
Thank you again for this great article, incl. The links to your conversations with a chatbot. I had no experience with that yet. No enthusiasm has arisen.
There are really a huge number of illusions where we also look.
Or am I too gloomy?
Is it full of debts a preconceived plan?
https://dissident.one/de-grote-diefstal-heeft-de-mondiale-elite-een-uitgebreid-plan-bedacht-om-alles-wat-we-bezitten-af-te-pakken
Is it believing in the happy ending that citizens have something to choose from an illusion?
https://www.youtube.com/watch?v=_7U5JVk_y7U
Is people responsible for a made -up climate problem?
https://maurice.nl/2025/08/16/de-verrassende-zoektocht-naar-de-verdwenen-hittegolven-uit-1947/
Are his road -made heat waves a symptom?
Or am I just too gloomy?
Lareb:
https://www.frontiersin.org/journals/drug-safety-and-regulation/articles/10.3389/fdsfr.2025.1644680/full
This seems strong to me when a failed attempt to knit straight ahead.
(P Koelewijn, apologies for the late posting: for some reason this comment ended up in the Trash, among hundreds of Russian spam messages. Perhaps unnecessarily, it was posted after all, for the sake of completeness.
Anton.)
See also Leon Kukler's findings about CIMS
This is stated in the article
Data on vaccination administration and data-linkage
At the beginning of the pandemic, no arrangements were present to establish the linkage with the national vaccination registry in the Netherlands (CIMS, maintained by the RIVM). Eventually, Lareb was granted access to this data through the RIVM. With the reporter’s consent, batch numbers and vaccine brands could be retrieved from the CIMS registry. This data was used to get the information in the SRS database as complete as possible. In addition, data from CIMS on the number of vaccinations administered in the Netherlands were provided by RIVM for signal detection activities, such as Observed vs. Expected analyses, where information on the number of vaccines administered—stratified by age and sex—is essential. Observed vs. Expected analyses became a standard analysis approach for COVID-19 vaccine signal detection, next to a clinical review of ICSRs, for events with a relatively high background rate.
It was not possible to link vaccination data with healthcare data in a fast and efficient manner in the Netherlands. This limitation hindered the timely evaluation of potential safety signals if needed. In contrast, many other EU member states were able to perform such linkages more effectively (Pottegård et al., 2021; Zureik et al., 2023; Ljung et al., 2021).
Are gems in it, such as:
“In contrast to the high number of reports on COVID-19 vaccines, the number of reports on drugs to treat SARS-CoV-2 symptoms was very low with only 265 ICSRs being reported. The high workload of healthcare workers in the middle of the COVID-19 pandemic has likely been an important barrier in reporting ADRs.”
"Important Barrier" My A ... Only in medicines and not in the vaccine?
Yes, they naturally wanted Bergen ICSRs from Ivermectin and Hydroxychloroquine, of those few unit who prescribed it and their job/big-region there. And fines risked.
Hepatitis E, according to RIVM Info 2023 there is only a vaccine in China. A lot of experience with hepatitis B, also with the puncture but hepatitis e has stayed out of my field of vision, so thanks for that "tip" after reading this article. At the beginning of 1981 I had an "ab o -like" experience when someone in my area would have received hepatitis B which later was in the file ... The doctor with a friend was so very happy with the patient that it felt embarrassing. The rest is history because the rollout of the Hepatitis B Vaccin did not take long. Unfortunately, very young babies already get this injected, see National vaccination program. What was there rather "the chicken prick" or "the egg disease"? Not with all the syndromes of course, but it seems more and more.
I see it in such a way that politics ordered ZSM to develop vaccines to stop the pandemic. Big Pharma finally sees a chance to sell mrna and says at some point: this is what it is. Politics decides that it is safe and effective and also has properties that have not been tested at all, for example prevent transmission. Those who are smart do not trust Pharma, I think the accountability is entirely with the "client". Then our government also knew that the surveillance systems would not be able to guarantee safety after the start of the mass vaccination.
About this scenario: https://virusvaria.nl/the-biggest-cover-up-in-history/
Dear Anton. What I don't understand is the following: At the end of February 2021, so 10 weeks after the rollout of the puncture, there were already 42,000 reports of side effects, of which more than 1,200 with fatal outcome. Source Pfizer. This had to be kept under the cap for 75 years, but the American Supreme Court put a stop to it. So all governments, including ours, were aware of this. Why do I don't hear anyone about that?
Probably because of the culprit that they are reports whose relationship with the vaccination has not been proven. And that there were a lot to do with the massive number of simultaneous pricks and the publicity that had been there.
In the meantime, I have spontaneously received a response from a reliable source that the calculations of both grok and chatgpt are not correct. This is despite the fact that both of them came out in the same ordering size. We'll find this out. In any case, the scope is not that it would be significant, on the contrary. It seems even worse.
And if it is the 10 largest vaccine trials how small than all other vaccine trials have been? As you write. So even more worrying
Since the EMA and the Health Council have settled with this relatively small sample, it implies that they advise positively on the safety of vaccines if there is no more than 1 person out of 1000 vaccinates as a result of the injection.
In my opinion, this is a far too large safety limit.
That should have been told to the population in advance.
And then some political parties also wanted to introduce a vaccination obligation based on these types of uncertainties.
Stupidity can be very deadly.
That mortality is of course also dependent on the target group. A trial at 20-30 year olds ultimately gives much lower mortality than during a trial at 60-70 year olds. To notice significantly higher mortality, the test group with lower ages will also have to be significantly greater.
I think I am herrining that mainly 18-50 year olds went into the trial of Pfizer.
Tests with whom people start are mainly healthy young men (except if the target group are pregnant women, for example, but then it is asked during a check and there is no compensation for it). The outrage that Pfizer, for example, did not test the C-Prick on pregnant women in the beginning is unjustified. That they should have stopped immediately after the first trial and everything that happened afterwards and still happens is very far beyond indignation.